
Chemical Energy Conversion
Small abundant chemicals such as N2, O2, CO2 and H2O can be converted into fuels or commodity chemicals via electrochemical processes.
Electrochemical Ammonia Generation
(EAG)
Each year, about 180 million tons of ammonia (NH3) are produced and transported worldwide. The current Ammonia production relies heavily on the energy intensive Haber-Bosch process. As a consequence, the NH3 industry is responsible for about 1.6 % of the world’s annual CO2 emission (420 Mt). Climate change and the imminent shortage of fossil fuel supplies demand a more sustainable and less carbon intensive way of ammonia production. Excess energy produced by renewable energy sources can easily be stored in the form of liquefiable fuels. Therefore, Electrochemical Ammonia Generation (EAG) has gained substantial interest as a viable replacement for the Haber Bosch process and as a new form of energy storage. The big advantage of this method over other energy conversion and storage systems is the world wide abundance of N2 and H2O, the two sole chemicals needed for EAG.

Electrical energy, generated by renewables can be converted into ammonia. The carbon free NH3 can easily be stored and transported.
At Hofkens Lab, we investigate new materials and setups for electrochemical ammonia generation. Our setups operate at ambient conditions, using only H2O as a proton source. A huge advantage of this approach is that the ammonia production could easily be delocalized (next to wind farms, solar fields or hydro stations).
The main focus of our research lies in finding new, stable electrocatalyst exhibiting a high selectivity and NH3 production rate. The as synthesized materials are characterized using state-of-the-art techniques. Promising electrocatalysts are also directly implemented in prototype electrochemical cells, proving the feasibility of the EAG process.

A standard electrochemical cell in operation. The cathode includes the newly synthesized electrocatalyst.
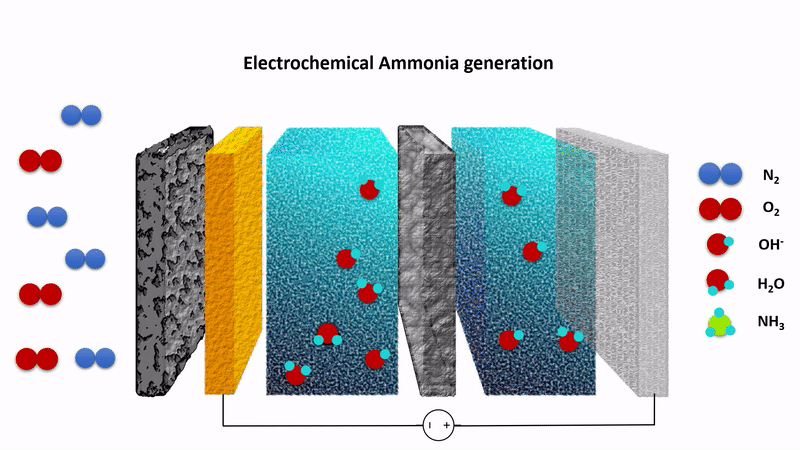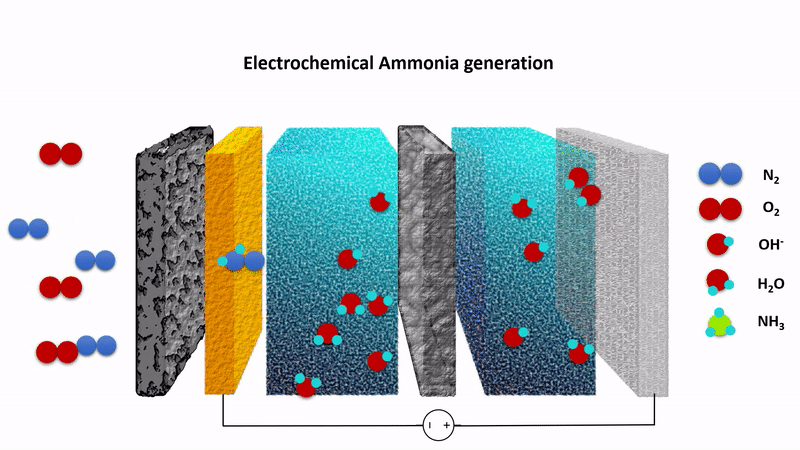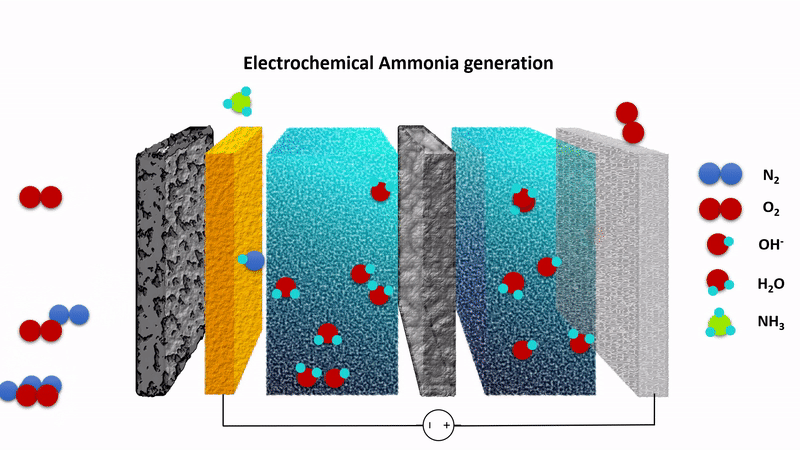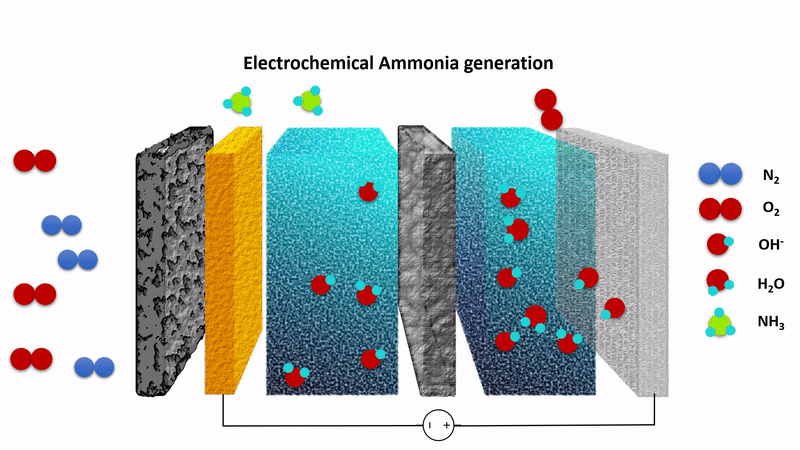
Animation showing the chemistry behind the EAG process. Nitrogen gas (N2) is converted to ammonia at the cathode (left) while hydroxide ions (OH-) are converted to oxygen at the anode (right). The process is driven by applying a fixed voltage between the anode and cathode.